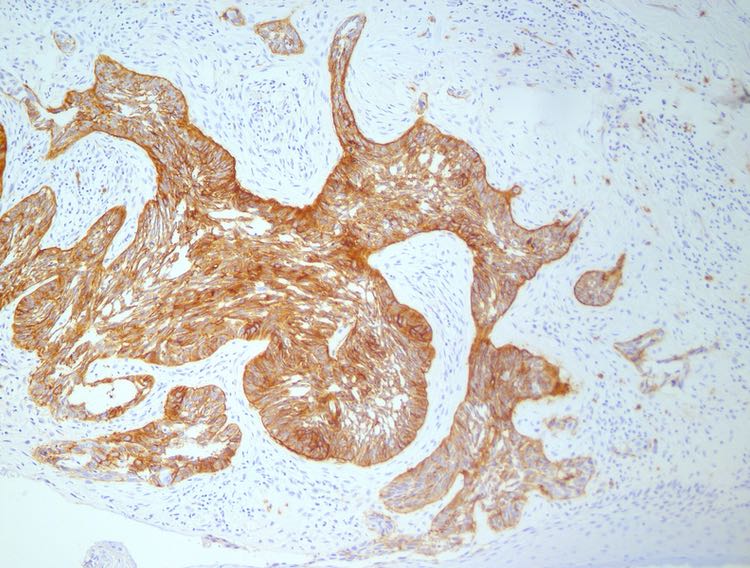
We are pleased to now share our additional positive findings following more detailed analysis of the clinical data. Despite this, the activity of Blautix relative to placebo in this Phase II study is competitive with approved IBS therapeutics. Topline results were significantly impacted by an unusually high placebo response in certain geographical patient groups. 'The Phase II results, in conjunction with regulatory guidance and KOL discussions, provide a clear and viable path forward for 4D pharma to continue to develop Blautix to address a significant unmet need. Alex Stevenson, Chief Scientific Officer, 4D pharma. We are encouraged by the positive outcomes of this additional analysis, and we strongly believe that this signal finding study supports the continued development of Blautix as a novel treatment for IBS,' said Dr. We have also discussed the results with internationally renowned key opinion leaders and patient groups. 'Following the announcement of the topline results in October, 4D pharma has been able to review the data in more detail. The clinically meaningful overall response rates and improvements in bowel habit in both IBS-C and IBS-D in this signal finding Phase II trial, in spite of an unexpectedly high placebo response in certain patient groups, are highly encouraging for subsequent larger studies with increased statistical power. Single strain LBP Blautix is the first therapeutic globally to have demonstrated efficacy in both IBS-C and IBS-D. In addition, analysis of the data by geographical region shows that earlier topline results were impacted by an unusually high placebo response in patients in the UK and Ireland, and enhanced positive signals were seen in the larger US population. Further analysis of the data has revealed strong and statistically significant activity on the key symptom of bowel habit, a potential approvable primary endpoint per regulatory guidelines. The data has been presented in a poster session at Digestive Disease Week (DDW), on May 22, 2021, by Professor Eamonn Quigley, M.D., Head of Gastroenterology and Hepatology at Houston Methodist Hospital, Professor of Medicine at Weill Cornell Medical, and Chief Investigator of the study.ĤD pharma previously announced topline efficacy and safety results from the trial, conducted in the US, UK and Ireland, in October 2020. 
Leeds, UK, 4D pharma plc (AIM: DDDD NASDAQ: LBPS), a pharmaceutical company leading the development of Live Biotherapeutic products (LBPs) - a novel class of drug derived from the microbiome, today announces additional positive data from its completed Phase II trial of LBP Blautix® in subjects with irritable bowel syndrome with constipation (IBS-C) or with diarrhea (IBS-D). New data demonstrating clinical activity in both IBS-C and IBS-D was presented at Digestive Disease Week 2021Ĭompany to host webcast on Tuesday, at 12:00 p.m. 4D pharma Presents Additional Positive Results of Phase II Study of Blautix® for the Treatment of Irritable Bowel Syndrome
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
